Gene Therapy 101: Fast Facts & FAQ’s
Written by Kristen Picard
Gene therapy. It’s a fascinating but complex nut to crack. If you’re interested in this topic but don’t have the time to scour the Internet for information, have no fear! In this post, I’ll give a brief introduction to this innovative field by answering some common questions.
Where did it begin?
The first gene therapy trials began more than two decades ago (Wirth, Parker, & Yla-Herttuala 2013). Currently in the United States, gene therapy is only available through clinical trials, with approximately 2000 trials initiated to date. In alignment with many new scientific advances, early research was not always successful; however, recent literature points to an incredibly bright future in gene therapy (Ginn et al. 2013, Naldini 2015, Shuvalov et al. 2015).
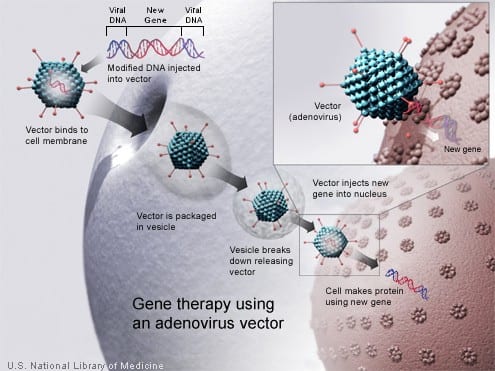
How does it work?
Gene therapy works by using a vector (which can be viral or non-viral) to introduce a new gene into a cell. The purpose for the new gene is to replace a defective gene or add a gene that supports cellular functions in a beneficial way. This two-minute YouTube video explains how vectors work in the body (here, the liver is the target organ): Gene Therapy Viral Vector Action.
Who benefits from these therapies?
Target diseases for gene therapy include those caused by faulty genes that lead to abnormal cellular functioning. Clinical trials using gene therapy are underway for cancer, diabetes, Cystic Fibrosis (CF), hemophilia, HIV, cardiovascular diseases, multiple sclerosis, myasthenia gravis, Alzheimer’s disease, Parkinson’s disease, inflammatory bowel disease, rheumatoid arthritis, renal disease, age-related macular degeneration, and blindness (as well as many other illnesses and injuries).
A few years ago, ABC released this six-minute news story about a young woman whose blindness was successfully reversed by gene therapy. There are hundreds, if not thousands, of stories like this.
What are the obstacles?
While gene therapy appears to be a promising treatment for many devastating illnesses, this development also comes with controversy.
Much good can be done through the application of this advancement, but there are fears about the exploitation of such an innovation. For example, there is the price vs. value discussion; how is the price determined for such a treatment? Also, will there be restrictions on the use of the application of genetic engineering in humans – will there be an open market for cosmetic usage, or the creation of “designer babies”?
While these are important considerations that will need to be addressed, let’s not forget the potentially life-changing nature of these therapies. I’m not sure if you feel the same, but for me, the consideration of the number of lives saved or improved by such therapies provides enough justification to continue research in this area.
I hope you enjoyed this intro to gene therapy. If you like learning more about new and interesting topics in biotechnology and other STEM subjects, be sure to follow us on Facebook and Twitter!
